ANGANY is a private, French-Canadian pharmaceutical company with mission to change our ability to fight allergy. Our goal is to help people with allergies get their life back.
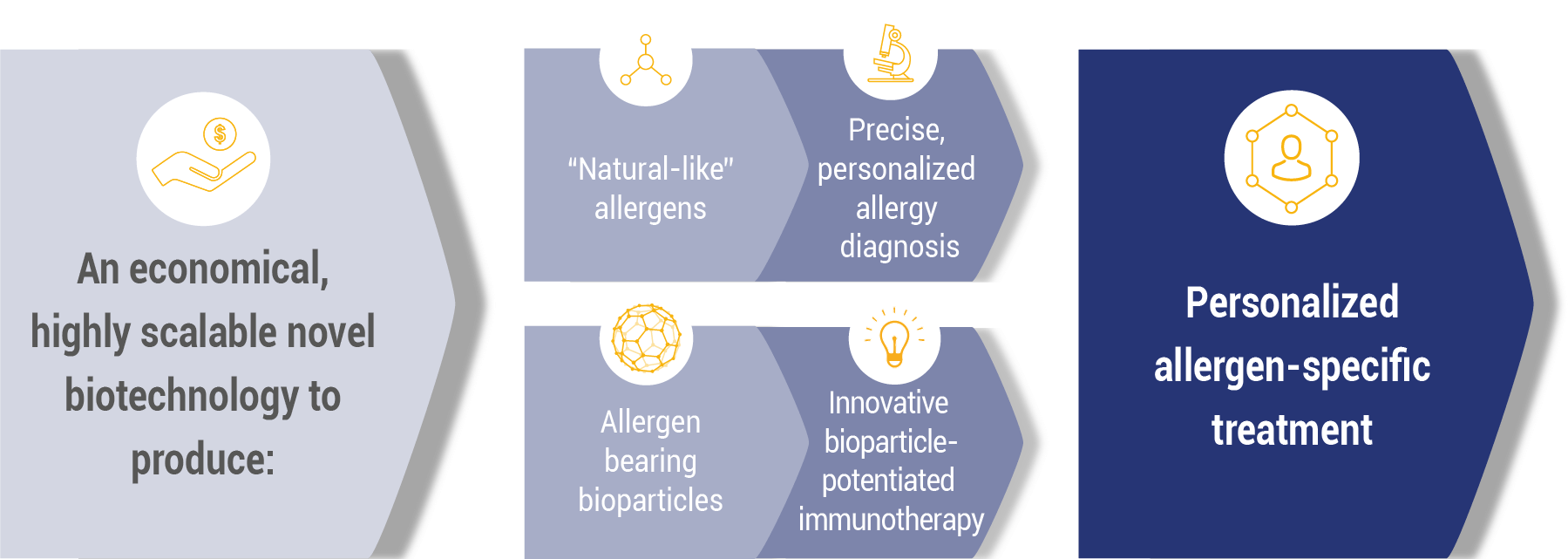
Leveraging 30 years of research in biotechnology, immunology and vaccinology, and after more than $ 15M of R&D investments, ANGANY has tackled the specific challenges of allergy to invent a new generation of pharmaceuticals. The goal: treatments worthy of precision medicine, simpler, more effective and safer than current desensitization, and based on a more precise diagnosis.
In a world first, ANGANY has developed an exclusive vaccine platform based on synthetic biology. This self-adjuvant vector, ANGANY’s proprietary eBioparticle ™, has demonstrated at the preclinical level the possibility of reversing the allergy dynamics. ANGANY has developed a flexible, fast and easy to scale 3rd generation plant-based production platform.
ANGANY thus wishes to bring lasting solutions into a reality where even today allergy sufferers must too often rely on drugs that only mask the symptoms of allergy. ANGANY has developed a form of immunotherapy that is simple in its administration, and from which we expect safety and long-lasting protective effects. Our goal is to stop the allergy dynamic that affects people and prevent the allergic disease from progressing.
ANGANY anticipates that its vaccine platform will also find direct applications in other therapeutic areas such as cancer, in the treatment of autoimmune, infectious and other diseases, in human and animal health.
ANGANY is a Canadian company founded in 2017 and which continues a mission started in France in 2010 by Dr Loïc Faye and Véronique Gomord and their biotech start-up Angany Genetics SAS.
ANGANY inc. is based in Quebec City; the company owns Angany Innovation SAS, an R&D laboratory located at the Pharmaparc de Val-de-Reuil in Normandy.